Why was Pfizer allowed to decide if a Myocarditis diagnosis was accurate?
To the best of my knowledge, there was no oversight of their assessment process. In turn, this significantly reduced their reporting requirements
Summary:
Pfizer used a recently defined Brighton Collaboration (BC) Level scale for myocarditis and judged that over 60% of VAERS reports “did not meet case definition”
This percentage directly contradicts what the CDC reported in its mid-2021 ACIP meeting. To be fair, they were not assessing the same set of reports, but that alone cannot explain such a large discrepancy
Pfizer likely did this to reduce the reporting requirements. Whenever Pfizer provided details about individual reports, it became quite easy to notice all the contradictions in their pharmacovigilance reporting
Foreign VAERS reports which were diagnosed as Myocarditis already met a pretty high bar. In my opinion, Pfizer should not have been allowed to reclassify them using their own BC Level classification system
Followup reports could add additional information and change a lower BC level to a BC Level 1. It doesn’t look like Pfizer used these followup reports to update their assessments.
I would like to revisit this chart from Pfizer’s submission to the European Medicines Agency (EMA)
There is something very strange about Pfizer’s classification numbers provided in the table above: almost 64% of VAERS reports (2624 out of 4106) were categorized as Level 4 - “insufficient evidence to meet the case definition”. If a VAERS report is categorized as Level 4, that means Pfizer was not sure if the vaccine injury was actually Myocarditis.
To give you a quick summary, this is what these Brighton Collaboration Levels mean:
BC 1 - definitive case (of myocarditis)
BC 2 - probable case
BC 3 - possible case
BC 4 - reported event with insufficient evidence to meet the case definition
BC 5 - not a case
Just to add some more context, here is how Pfizer describes these levels in one of their pharmacovigilance reports:
The definitions for these levels are discussed in more detail in this paper which was published primarily to establish Myocarditis diagnosis for COVID19 vaccines1
These percentages do not even match CDC’s own numbers!
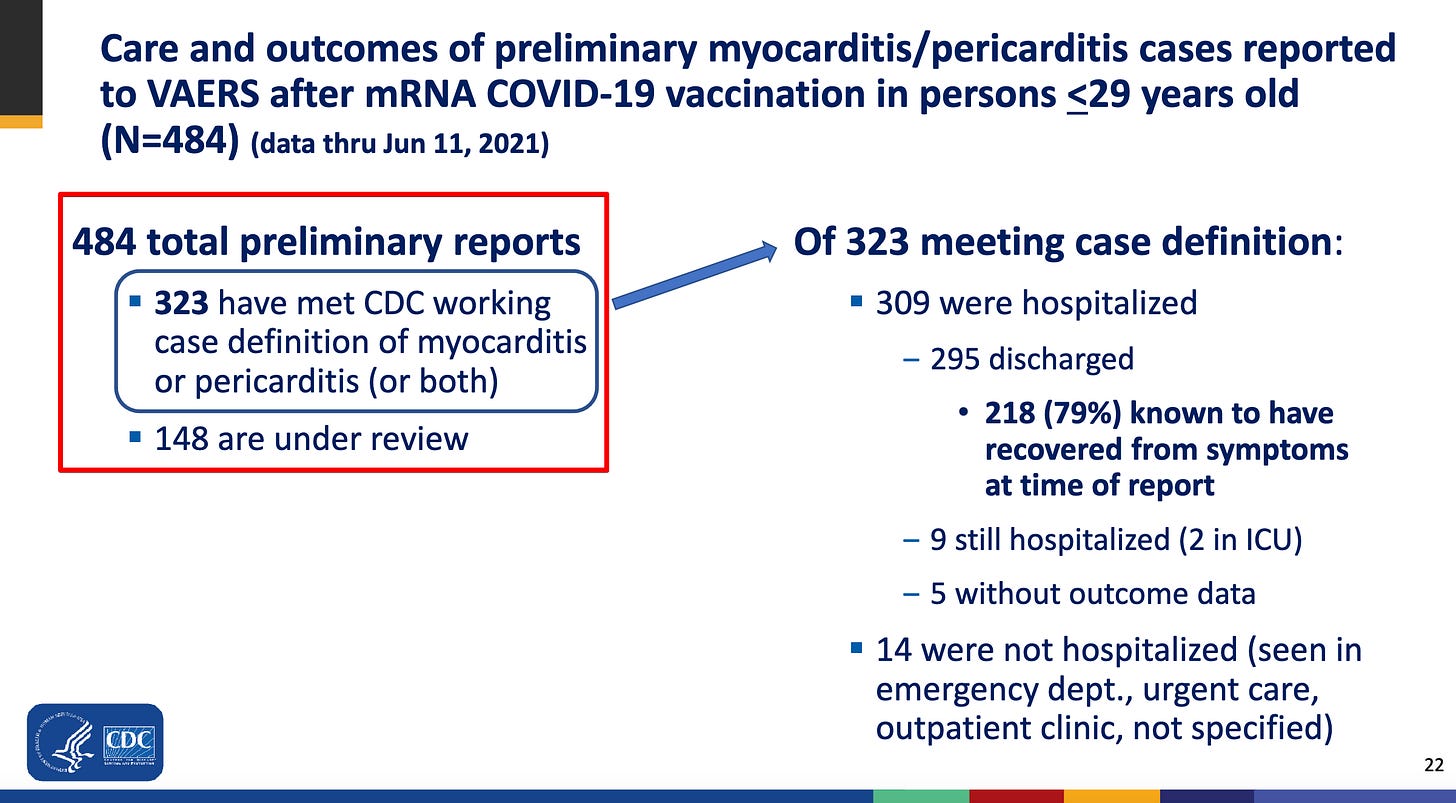
As a contrast, here is a slide from an ACIP meeting from mid-2021:
As you can see, from 484 preliminary VAERS reports, 323 met the case definition (that is, the CDC judged that the vaccine injury was actually Myocarditis)
This gives us a percentage of 323/484 = 66%
To be a bit more objective, it is important to mention that the CDC was only considering the age group under 30, while Pfizer was considering all age groups.
But still, this is practically an inversion of case vs non-case percentages between CDC and Pfizer!
Pfizer had an incentive to classify reports as non-cases
In the same EMA document, we also see this verbiage:
We note that for myocarditis only BC level 1 cases are primarily reviewed. However, for future reviews, we find it important that also BC level 2 cases are closely considered in the analysis, as cases assessed as BC level 2 add valuable information regarding myocarditis risk after vaccination.
In other words, not only did the EMA want level 1 cases to be reviewed (that is, they expected Pfizer to provide details for individual reports), they also wanted Pfizer to do the same for Level 2 reports.
This incentivized Pfizer to classify reports as low as possible on the Brighton scale because it obviously reduced their reporting requirements.
Why do these reporting requirements matter?
Less reporting requirements = less chance of self-contradiction
Somewhat comically, the only time that Pfizer mentioned individual VAERS report numbers (to the best of my knowledge), I was immediately able to spot a few contradictions in their pharmaco-vigilance reporting!
In fact, even without the individual VAERS report numbers, sometimes it is possible to find them just from the case description provided in these documents.
For example, I was able to find the corresponding VAERS reports based on these descriptions:
As you can see, Pfizer would want to avoid discussing individual cases as much as possible.
On top of that, by pushing all the myocarditis VAERS reports down to the lowest possible Brighton Collaboration level, they also persuaded the EMA to write this in the same document (emphasis mine):
A minority of cases (10%) qualify for BC level 1 classification, and thus with a high degree of diagnostic accuracy. Even in these cases where myocarditis or pericarditis diagnosis is confirmed, causality assessment was not possible for most of these cases due to lack of proper accounting of case duration, severity, outcome, concomitant medications and/or investigative measures to exclude alternate aetiologies such as viral infections, cardiovascular disorders. These limitations of the post-marketing data are important factors that preclude proper medical assessment of causality between the event occurrence and vaccine administration.
In other words, the EMA would like us to believe post-vaccine Myocarditis stuff isn’t really a big deal.
Unfortunately for Pfizer,
a) someone decided to sue them for all the documents they submitted as part of the pharmacovigilance
b) and VAERS makes it fairly easy to spot all the internal contradictions in the pharmacovigilance reporting2
No wonder the vaccine pushers are furious that people are analyzing VAERS!
Who files foreign VAERS reports?
A few years back, the CDC published this (emphasis mine):
During 2011-2014, VAERS averaged around 30,000 U.S. reports annually, with 7% classified as serious. Healthcare professionals submitted 38% of reports, vaccine manufacturers 30% and patients and parents 14%. Reporter type and percent of serious reports vary across vaccines, age of vaccine recipient and how long the vaccine has been in use. During this same time period VAERS averaged around 6,000 foreign source reports annually. Vaccine manufacturers, which accounted for >99% of foreign source reporting, are required by law to submit foreign source adverse event reports that are both serious and unexpected [21], but not other types of foreign source reports. Given the vaccine manufacturer reporting requirements and the minimal amount of direct public reporting, it is not surprising that a relatively high percentage (48%) of foreign source reports are classified as serious. This likely represents selective reporting based on regulatory requirements rather than any substantial differences in safety profiles of foreign vaccines
Notice that the verbiage specifically says serious AND unexpected.
Here are the definitions of serious and unexpected:
And on average, nearly 70% of serious foreign VAERS reports are filed by healthcare professionals.
Identifying REPORTER field from the writeups of serious VAERS reports
People may not know this, but VAERS actually collects a lot of information about the person who files the report. But they don’t publish the information collected within the checkboxes. (To the best of my knowledge they don’t even provide aggregate information about the Reporter checkbox, if I am wrong please leave a comment).
So what does this mean?
Personally, I think Pfizer should not even be reclassifying foreign VAERS reports into Brighton Collaboration levels3, because a vast majority of them were filed by healthcare professionals!
Given that the healthcare professional who treats the patient has first hand knowledge of not just the symptoms but also likely the patient’s history, why was Pfizer allowed to overrule the diagnosis of healthcare professionals?
In other words, if a foreign VAERS report has been diagnosed as Myocarditis, there are two possibilities:
not all details were provided in the VAERS report, but the injury did meet the case definition of myocarditis
all details were provided in the VAERS report, and the injury did not meet the case definition of myocarditis
Given that filing a VAERS report completely (or any online form, really) can be quite tedious and error-prone, I believe Option 2 is much more likely than Option 1
Update: I meant to write Option 1 is much more likely than Option 2 :-) Sorry about the confusion!
Followup reports add to the complexity
I have written a series of articles about followup reports, and explained how they often add new information to the original report.
Why deleting followup reports automatically understates vaccine danger signals
Some recent comments by readers prompted me to look into the quality of deleted VAERS reports. We now know that the reason for the deletion is not nefarious (at least I don’t have any reason to think so till now) - they keep the original reports but delete followup reports.
This implies that followup reports for Myocarditis injuries could provide additional information which would allow the assessment to become BC Level 1, even if the original report was at a lower level.
As best as I can tell, it looks like Pfizer did not use the information from followup reports to update their assessments. They certainly don’t mention using followup reports anywhere in their pharmacovigilance documents4.
Conclusion
To the best of my knowledge, Brighton Collaboration Level assessments were not directly used in risk-benefit analysis calculations. (Please let me know if I am wrong about this).
However, by asking Pfizer to do its own assessments of the BC Level for these injury reports, the health regulators allowed them to get away with much less stringent reporting requirements.
While this may have created an illusion of a thorough and diligent process, there were actually a lot of contradictions in Pfizer’s pharmacovigilance reporting.
If you are interested in following a somewhat humorous rabbit hole on how this “paper” was peer reviewed, you can read my Twitter thread
This is why I think the CDC is going to cripple or even entirely shut down VAERS before they attempt another mass vaccination rollout. They have already done this to the EU VAERS reports
Unfortunately I cannot say the same about US VAERS reports. It does look like a lot of US VAERS reports are filed by consumers. While that makes the US system more open and fair, consumers are not trained on using VAERS and hence leave out a lot of important information
Which does make sense if you are Pfizer. Why open yet ANOTHER Pandora’s box? :-)










VAERS/Shimabukuro also used the BC to evaluate anaphylaxis. This enabled them to exclude much of the anaphylaxis from the reporting set. https://jamanetwork.com/journals/jama/fullarticle/2776557 When you dig through this "report" it tells you that they used the BC to evaluate reports. In BC Anaphylaxis this is a 1-5 scale. They eliminated level 4 and 5 basically eliminating any report that said Anaphylaxis but gave no details (whether or not it was made by a HCW). It also meant that if anything was miscoded in the symptoms that was not considered either. I have one report in my slide deck that calls a clear case of Anaphylaxis---- wait for it.... Tongue Disorder... There are thousands of reports in VAERS where Anaphylaxis is clearly misclassified as tongue issues, throat swelling, itchiness etc... here is just one example: https://www.openvaers.com/vaersapp/report.php?vaers_id=903324 This is why OpenVAERS has an "anaphylaxis" number (currently 10+k) and a Severe Allergic Reaction number (currently 46+K).
9th century - the first medical school in the Western world opened in Salerno, Italy (Schola Medica Salernitana)
1895 - the first heart surgery performed (Oslo)
1979 - the first successful heart transplant (Cape Town) (“The surgeon who dared” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6062759/)
2023 - we still don’t know what “heart failure” means…