A closer look at the <0.1% v-safe hospitalization rate claimed by CDC
And why the free text entries matter
Summary:
I have created a tool to visualize the timeline for v-safe registrant checkins
There are many issues with the 0.1% hospitalization rate in the CDC v-safe paper published in the Lancet
a 0.1% hospitalization rate is still very high
about 20% of the registrants in the data subset I considered were only hospitalized after Dose 3. By considering only Dose 1 and 2 in their paper (even though the information about future doses had been collected), the CDC ignored a large amount of data in their analysis. I have included information from Dose 3 and Dose 4 checkins in my analysis.
the paper uses a risk window of 7 days, while most vaccine safety studies use 42 days. This does make a big difference to the number of hospitalizations.
even with a 42 day risk window, it is possible people who get hospitalized after Day 42 had initial symptoms earlier (and there are many such examples in the v-safe data)
In its highly cited Lancet paper, the CDC claimed that less than 0.1% of v-safe registrants reported that they were hospitalized.
This claim is highly misleading (because it cherry picked an incomplete subset), and I have now built a v-safe visualization tool so people can verify what I am discussing in this article.
v-safe visualization tool
I sometimes hear about people doing research into v-safe data, but it is quite clear to me that it is too hard to really do any proper analysis of the data without a tool to visualize it in the first place. v-safe is a uniquely complex dataset1 - so why has no one2 spent the time to build a visualization tool in the first place?
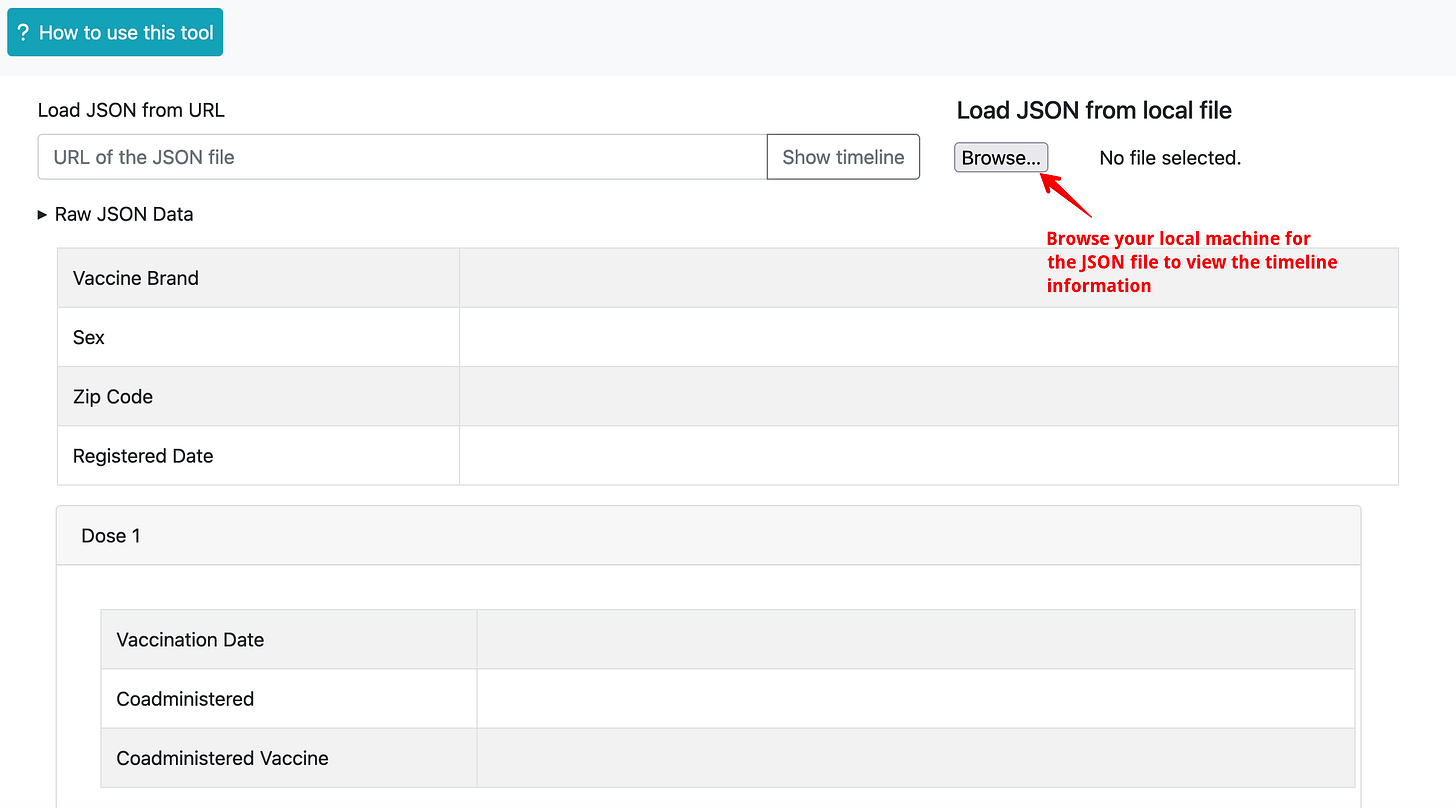
You will need to get a URL of a JSON file which contains information about a given registrant and paste it into the textbox and click on “Show timeline” button
Alternatively, you can download the same JSON file to your local machine and click on the “Browse…” button and open the file and see the timeline.
Where do you get the JSON file?
I will usually provide a link to the JSON file where possible, or I will provide a link to a zipped folder which contains the JSON file.
In the former case, you can just paste the URL into the textbox and see the timeline info. In the latter case, you can select the JSON file (whose filename will match the registrant ID) from your local machine and see the timeline information.
So let us now move on to the discussion of the primary topic.
A 0.1% hospitalization rate is still very high
So the first thing which is truly striking (at least for me) is that a 0.1% rate of hospitalization is still WAY TOO HIGH and must have been flagged for further research by the paper reviewers.
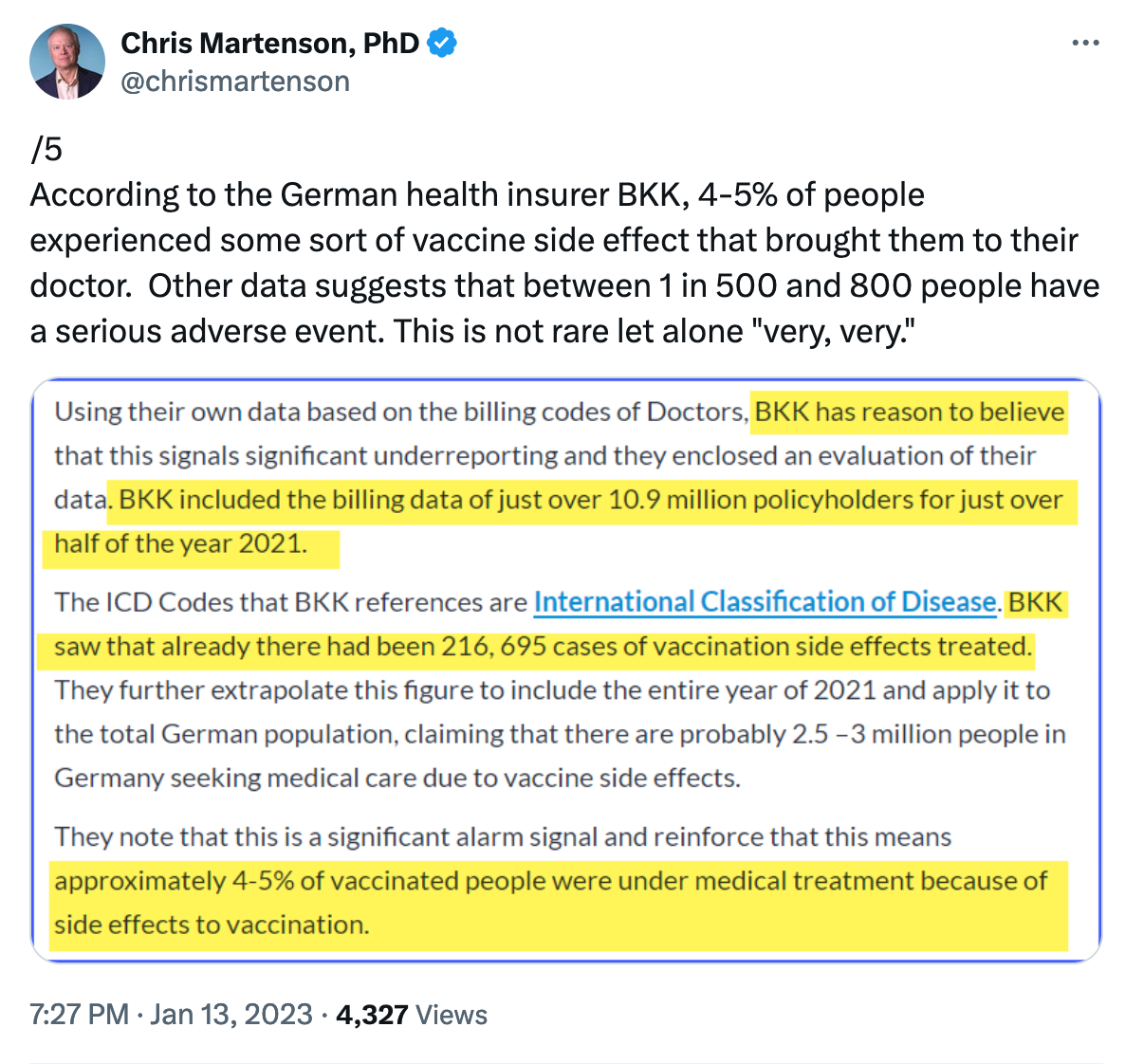
This is about 1 in 1000 people, which does closely match some data we received from a German health insurer in early 2023.
Most vaccine safety analysis uses a risk window of 42 days
Most of the safety analysis for the COVID19 vaccines use a risk window of 42 days. And this is in fact very easy to do using v-safe, because they already track the patients on the following schedule.
Daily for the first week:
Once a week for the next 5 weeks, until Day 42, and then checkin after 3 months, 6 months and 12 months.
But the Lancet paper only looks at the hospitalization rate for the first 7 days to calculate their <0.1% number.
Maybe the participants were selected using this rule, but did they expand the risk window of their analysis to a larger number, like 42 days?
The Limitations section clarifies:
“Finally, this report only included v-safe responses received during the first week after vaccination.”
And what does this mean?
a) given the importance of this paper - to the best of my knowledge, this was the only paper that the CDC published on the topic of v-safe which provided all this aggregate analysis
b) and the fact that v-safe does collect data for an entire year, and a lot of people had completed the full year of checkins by the time this paper was written
c) and the lack of systems external to the CDC which people can use to verify the paper claims3
this indicates a non-existent or at least a very shallow peer review process, and entirely contradicts Dr Dan (Debunk the Funk) Wilson’s opinion (from the video cited in the previous article) that “This is what scientists do. They ask a question and then attack it from multiple angles to see if the evidence yielded from multiple angles are all consistent with each other.“ Ha ha… Well, obviously not!
So how much of a difference does a 42 day risk window make compared to a 7 day risk window?
Consider the Vaccine Enthusiast Pfizer Cohort4
There are 1297082 registrants in this subset (download the full list as CSV).
I have created a dataset for the registrants within the “Vaccine Enthusiast Pfizer Cohort” who reported in one of their checkins that they were hospitalized.
There are 8751 such registrants.
A 0.1% rate of hospitalization means we expect only about 1300 people to be hospitalized. While not all the 8700+ registrations were due to the vaccine, I will explain why the number is still more than the sub-0.1% that the CDC claims in its paper.
Hospitalization after Dose 3
There are a lot of people in the dataset who were hospitalized only after their 3rd dose.
You can find them by using EARLIEST_HOSP_DOSE greater than 2.
This is almost 20% of the total number of hospitalizations in this data subset (VPEC), which means the CDC probably missed out on 20% of the hospitalizations by ignoring Dose 3 and later.
Given that the denominator - the number of people who received Dose 3 and 4 - was said to be substantially smaller than the number which received the first two doses, this has an additional implication: this means it is possible that the 3rd and 4th dose increase the risk of an adverse reaction compared to Dose 1 and 2. By ignoring this information, the CDC prevented us from learning about some important information.
The rest of this analysis also includes v-safe checkins for Dose 3 and 4.
Extending the risk window to 42 days
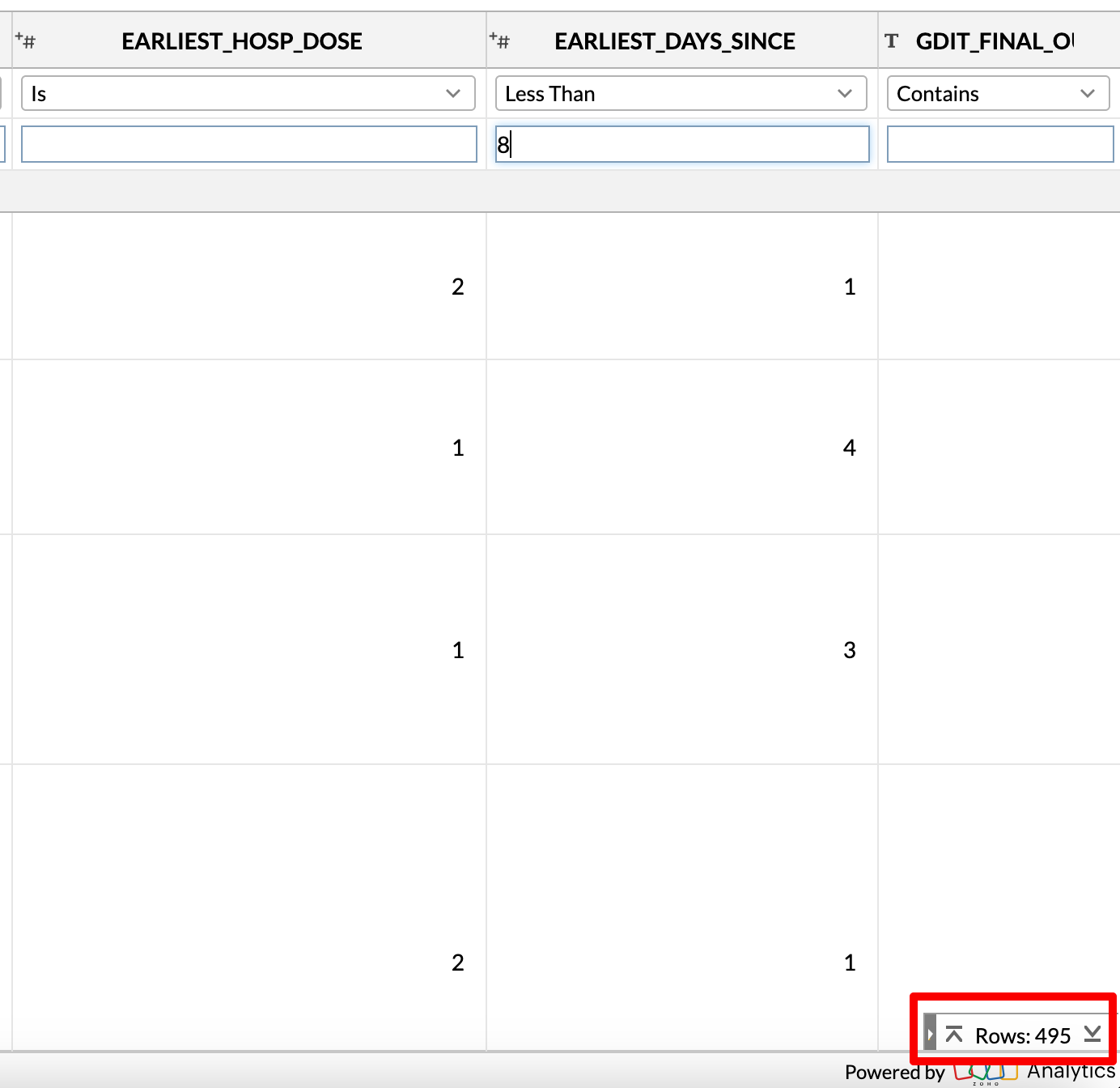
I added some code in my Python script to calculate the earliest dose for which they reported being hospitalized, and the earliest number of days it happened after their most recent vaccination (based on the SURVEY_STATIC_ID).
When I filter for EARLIEST_DAYS_SINCE < 8, there are only 495 registrants who make the cut.
If I increase the risk window to 42, almost 2500 registrants make the cut. (This is almost 0.2% of the cohort size, almost double what the CDC claims).
Not only is this a substantially higher number, it also suggests that the following claim made by Dan Wilson is almost entirely wrong.
So if you look at the V-safe data, the greatest number of reported events occurred on day one following the dose of vaccine being given.
[00:11:01]
From there, the average number of reported events declines each day. So even if they're just looking at the first seven days, you're capturing most of the reactogenicity that is happening with these vaccines.
People who get hospitalized after Day 42
But there is actually a problem with even the 42 day risk window, which we can infer using v-safe data itself.
If you have been following this Substack for a while, you might remember how CDC simply does not publish followups to VAERS reports for people who experienced severe adverse events, even if the followup is actually a death report.
How the VAERS followup process hides death reports
Someone left this comment on my previous article:
The BMJ even wrote an article which touched upon this:
Our investigation has also found that, in stark contrast to the US government’s handling of adverse reaction reports on drugs and devices, the publicly accessible VAERS database on vaccines includes only initial reports, while case updates and corrections are kept on a separate, back end system. Officials told The BMJ that this was to protect patient confidentiality—but this means that patients, doctors, and other public users of the database have access only to an incomplete and uncorrected version.
Someone responded to the article in the comments section, defending VAERS (emphasis mine):
Despite criticisms highlighting its limitations, VAERS's crucial role in vaccine safety shouldn't be overshadowed. It is a part of a comprehensive safety strategy, working alongside VSD, CISA, and V-safe. These collaborative efforts ensure a robust approach to vaccine safety monitoring. The key responsibility is to engage thoughtfully with VAERS, ensuring vaccine safety and maintaining public trust. Educating the public about VAERS and fostering scientific literacy are essential for upholding public health standards. Responsible use of VAERS data, in conjunction with other systems, is crucial for effectively navigating public health complexities and ensuring the integrity of vaccine safety monitoring.
But if longer risk windows are not utilized by the CDC when they write papers about v-safe, then there is nothing “comprehensive” about the analysis.
Why make tall claims about long term monitoring of vaccine injuries if you are not going to actually use all the data which has already been collected?
In the case of v-safe, because the app does follow up for a full year, it is possible someone experiences a severe reaction within the first 42 days, and ends up getting hospitalized only after the 42 day risk window.
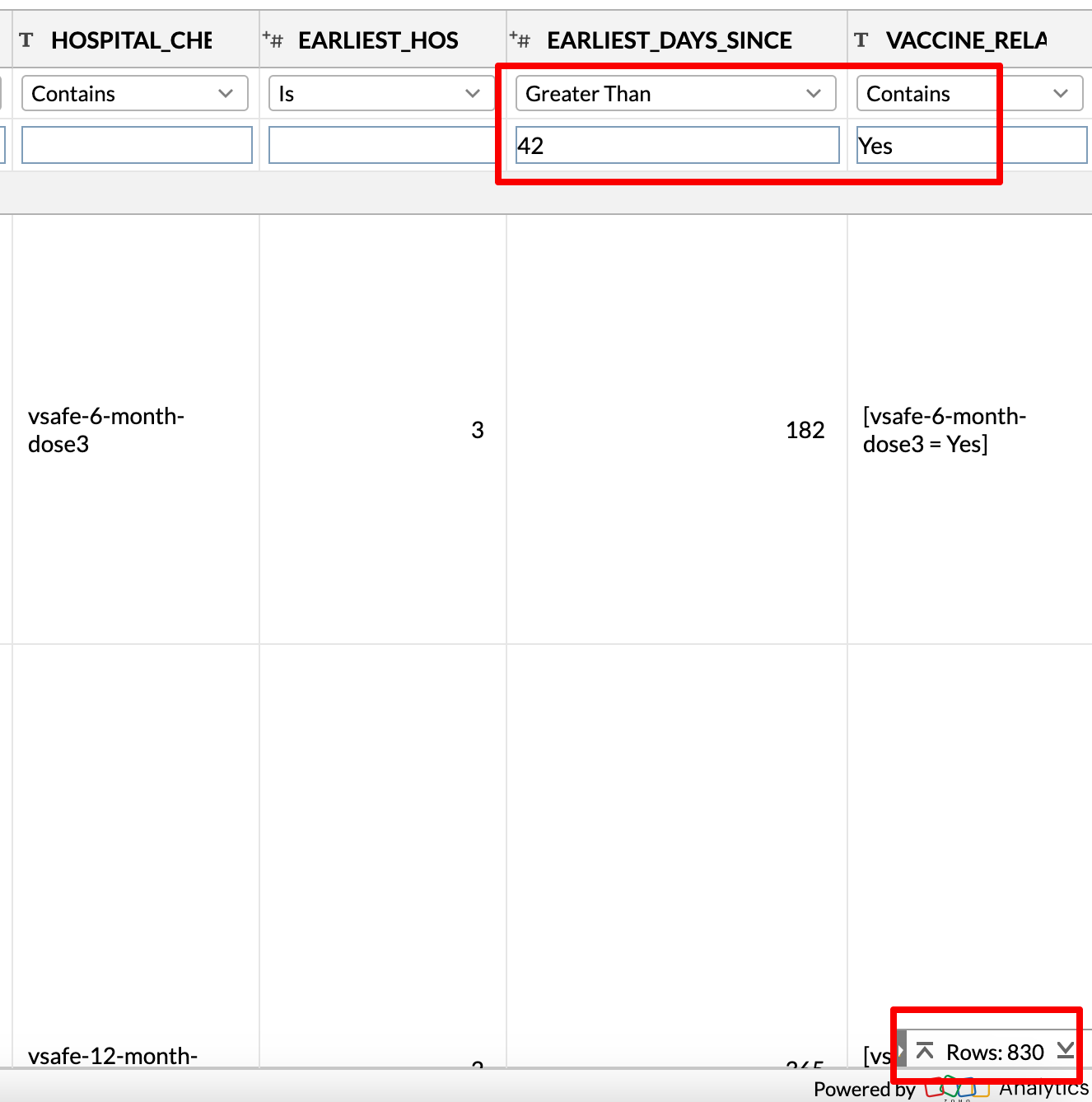
You can find these people by filtering for EARLIEST_DAYS_SINCE > 42 and VACCINE_RELATED contains ‘Yes’. The VACCINE_RELATED field is based on whether the user answers ‘Yes’ to the following question: ”If worse, do you believe your health problems might be related to your COVID-19 vaccination?”
There are 830 such registrants.
These are people who believe that the vaccine caused their health issue5, but the hospitalization occurred more than 42 days after their most recent dose.
In fact, you can narrow this down even more using the MEDICAL_CARE_CHECKINS field. This field simply lists all the survey ids where the user reported that they received some medical care (including things like telehealth consultation).
Suppose you filter for the word “daily” in MEDICAL_CARE_CHECKINS - there are 30 such entries.
Consider registrant 22K-12043-63994. (Note: the link is to a JSON URL, which you can plug into the v-safe timeline tool).
She had an initial reaction to Dose 2 in the first week:
But was only hospitalized by the time of the 3-month checkin
This will not be captured by the 42 day window for hospitalization, but it is highly likely that this hospitalization occurred as a result of the initial adverse event.
Why analysis of free text entries matters
Out of the 8750 registrants, 2400+ filled out free text entries. In other words, more than 1 out of every 4 people who got hospitalized filled out a free text entry.
Not all of it is useful or relevant, but how would the CDC know without doing any analysis?
What kind of text analysis did they actually do?
And why didn’t they share this free text analysis with the public?
Because even though nearly all the data is relational (i.e. queryable using something like SQL), you need to see the full timeline to get the complete picture, and a multi-dose vaccine schedule makes this timeline view more complex. In fact, as I was writing this article, I caught a fairly major error in my analysis because I was able to visualize the data.
Obviously I am not referring to the independent researchers who are doing all this work completely on their own dime. I am talking about people who claim to have large teams working on this research. Who is leading these teams? Do they have the necessary skillsets? Why is no one able to get some kind of an open source v-safe project started, soliciting contributions for building these kinds of tools? In fact, a well constructed user interface can even be used to crowdsource the free-text analysis. Why hasn’t anyone from these large teams done any of this groundwork?
That is, we are being asked to take the CDC at their word, and the people who are supposed to be the first line of defense - aka paper reviewers - don’t seem to have access to the underlying data
These are the people who reported feeling “Fine” during their first checkin, and also did not report any symptoms. They are “vaccine enthusiasts” because they did not report to v-safe because they had an injury, but rather they volunteered to help out with the data collection. Of these vaccine enthusiasts, I consider the cohort where people got only the Pfizer vaccine for each of their doses and I refer to it as the Vaccine Enthusiast Pfizer Cohort (VEPC) in my writing.
This does not necessarily mean it is true. But in that case, I hope the vaccine pushers would also agree that just because someone leaves the field blank does not mean it is false.














Do you know, what happens if a v-safe participant dies? Or is injured to the point of not being personally able to follow up? I am guessing they simply drop off the radar.
Excellent article by the way.